Flame Retardant Analytical Standards
Use and Concern over PBDEs
Polybrominated diphenyl ethers (PBDEs) are flame retardant chemical additives to polymers (plastics) that are used in the manufacture of printed electronic circuit boards, clothing, drapery, upholstery, cushioning foams for furniture, batteries, automotive interiors, boats and many other consumer products. The flame retardant activity is a result of the effective electron-scavenging action of bromine. PBDEs comprise a large number of congeners that are often used in combinations; the choice of individual congener or blend depends on the polymer formulation and its end use.
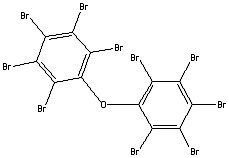
Figure 1.Decabromodiphenyl ether. Flame retardant still permitted under current EU regulations.
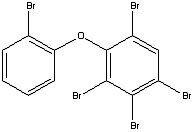
Figure 2.Pentabromodiphenyl ether. A EU-banned flame retardant substance.
Although their life-saving benefits and value as flame retardants are indisputable, leaching of PBDEs1 and workplace exposure pose environmental and health threats2. Exacerbated by their ubiquitous use, they are persistent organic pollutants prone to bioaccumulation. Measured levels of PBDEs in human tissue have been doubling every 4 to 5 years since 1970. Studies have shown high PBDE levels in neonatal blood matching the levels found in the mother’s blood3, and substances related to Penta-BDE have been found in breast milk and other tissues4. Besides concern over the effects of PBDEs on humans, other studies show that there may be additional environmental dangers, including causing thin shells in Peregrine falcon eggs5. Although their specific health consequences have not been fully elucidated, they are similar structurally to thyroid hormones6 and exhibit a wide spectrum of toxicity in laboratory animals7.
Risk Assessment
Many PBDE-based flame retardants have been phased out, but because of the importance of PBDEs to both manufactures and consumer safety and the lack of ideal alternatives, it was deemed unwise to eliminate them completely as a class without further study. To date, all commercial PBDE products have undergone evaluation under the Existing Substances Regulation 793/93/EEC8. Based on this risk assessment, the EU has banned the use of Penta- and Octa-BDE, but permitted Deca-BDE. A summary of progress is available on the Web site www.cefic-efra.com under “Library”. Summarizing9:
“Penta-BDE and Octa-BDE. For both these products, the Risk Assessments concluded that their use presented no risk to consumers’ health through contacts with treated products, but raised questions concerning possible effects on industry workers handling the pure chemical and concerning possible bioaccumulation. Therefore neither of these products is used today in the EU.
“Deca-BDE. After ten years of extensive studies, the final Risk Assessment was completed in 2004 and confirmed in 2005. The study concludes that the use of the product presented no risk for human health, neither through consumer contact with treated products, nor to workers using the product in industry, nor for indirect exposure of the population via presence of the product in the environment. Also, the report concludes that the product presents no identified risk for the environment, via air, water, soil, sediments or sewage sludges, but that environmental monitoring and further studies should be continued.”
Regulations
The primary regulatory methodologies regarding the analysis of PBDEs include:
- EPA Method 527. Determination of Selected Pesticides and Flame Retardants in Drinking Water by Solid Phase Extraction and Capillary Column Gas Chromatography / Mass Spectrometry (GC/MS). Includes, besides various pesticides, PBDE-47, PBDE-99, PBDE-100, PBDE-153.
- ASTM Method 1614. Brominated Diphenyl Ethers in Water, Soil, Sediment and Tissue by HRGC/HRMS. Eight congeners of primary interest are listed: PBDE-28, PBDE-47, PBDE-99, PBDE-100, PBDE-154, PBDE-153, PBDE-183, PBDE-209.
- ISO 22032:2006. Water quality. Determination of selected polybrominated diphenyl ethers in sediment and sewage sludge. Method using extraction and gas chromatography/mass spectrometry. PPBDE congeners determined: PBDE-47, PBDE-99, PBDE-100, PBDE-154, PBDE-153, PBDE-183, PBDE-209 (does not include PBDE-28).
Reliable Analytical Methods using our Products
Underlying the ongoing deliberation over the regulation of PBDEs is the need for reliable analytical methodologies. Three components are important to a successful analysis of PBDEs:
- PBDE standards
- Solid phase extraction tubes
- Capillary GC columns
We carry all of the necessary components. Recently, we completed our offering of PBDE standards by introducing sixteen new congeners (Table). Shown in Figure 3 is the GC-MS analysis of fourteen PBDE compounds on a Supelco SLB-5ms capillary GC column, which is ideally suited for this sensitive analysis.
Continue to look to us for high quality, relevant products to meet today’s most pressing analyses.
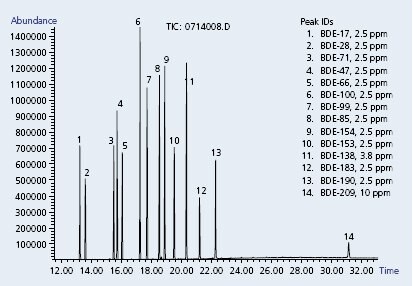
Conditions: Capillary GC column: Supelco SLB™-5ms, 30m x 0.25 mm I.D. x 0.25µ (Cat. No. 28471-U); Oven: 125 °C (1 min.), 10 °C/min. to 340 °C, Scan range: SIM; Carrier gas: Helium, 1.5 mL/min., constant; Injection: 1µL, splitless, pulsed (30 psi until 0.2 min.); Liner: 4 mm I.D. single taper.
Sample: PBDE standards, 2.5-10 µg/mL
Product Listing. PBDE Standards
(Ampuls of 50 µg/mL PBDE standard in isooctane, except as noted.)
References
For Additional Information
BSEF – Bromine Science and Environmental Forum: www.bsef.com
EFRA - European Flame Retardants Association: www.cefic-efra.com
To continue reading please sign in or create an account.
Don't Have An Account?