Step-by-Step Guide for KitAlysis™ Suzuki-Miyaura Cross-Coupling Reaction Screening Kit

- 1 mylar bag from the KitAlysis Suzuki-Miyaura Reaction Screening Kit and you will use the following components
- 6 x 4 pre-weighed catalysts in glass vials loaded with stir bars and topped with cap mat
- 4 empty 4 mL substrate stock solution vials - 1 NEW KitAlysis 24-Well Reaction Block Replacement Film
- 1 (2mL) ampule each of Toluene, THF, n-Butanol, DMAc, and aq. 1.5 M K3PO4 (in ampule boxes)
- 4 NEW stir bars
- KitAlysis 24-Well Reaction Block (sold separately)
- KitAlysis Benchtop Inertion Box (sold separately) or glove box/glove bag
- KitAlysis Torque Screwdriver Set (sold separately)
Additional (not included) items needed:
- Pipette (0-100 µL) & tips
- 4 (1 mL) syringes with long needles
- Your (het)aryl halide and (het)aryl boronate
- Nitrogen (or Argon): from hood line or tank
- 1 hot plate with stir capabilities
- 1 stir plate (or hot plate to be used without heat)
- HPLC vials, 96-well HPLC auto sampler block, or TLC plates
Set-Up Procedure
- Preheat a hot plate to 60 °C (use oil bath or second reaction block to hold temperature and avoid spiking). Reaction temperatures for the Suzuki-Miyaura reaction can range from room temperature to 120 °C but 60 °C is a general and very good starting point.
- Place a NEW KitAlysis 24-Well Reaction Block Replacement Film on reaction block lid (make sure all holes, including the temperature probe hole, line up with the corresponding holes on the film).
- Check all screws to ensure they are not stripped. Replace any stripped screws with provided replacements.
- Place the KitAlysis Benchtop Inertion Box with tubing connected to inert gas onto the second, non-heated stir plate (see KitAlysis Benchtop Inertion Box set-up for details).
- Place KitAlysis 24-Well Reaction Block with lid into KitAlysis Benchtop Inertion Box. Start nitrogen flow and purge 5 minutes. Leave nitrogen flowing for remainder of set up.
- Weigh both substrates directly into all of the empty 4 mL reaction vials according to recipe (provided in the downloadable excel file) omitting solvent and base solution. Add one stir bar to each vial mixture. Label as “DMAc Substrate Mixture A”, “Toluene Substrate Mixture B”, “n-butanol Substrate Mixture C”, and “THF Substrate Mixture D”).
- Partially open the lid on the KitAlysis Benchtop Inertion Box. Place the “DMAc Substrate Mixture A” and the “Toluene Substrate Mixture B” in two of the holes located on the left hand side of the Inertion Box diffuser tray. Place the “n-butanol Substrate Mixture C”, and “THF Substrate Mixture D” in two holes on the right hand side of the plate. Ensure that one vial is placed in the center hole on either side. This vial placement allows for the best flow of inert gas (remove lids from the solid mixtures before placing them into the recommended holes, keeping the lids in the Inertion Box for later use if needed).
- Transfer the capped, 24-vial, preloaded catalysts into the reaction block making sure to load it according to the matching diagram (A1:top Left corner) on the packaging and the Reaction Block. Leave the cap mat on.
- Using an ampule cracker, open 1 ampule each of DMAc, Toluene, n-butanol, and THF and quickly place into ampule holes located along the bottom of the Inertion Box, below the Reaction Block. Then open 1 ampule of the base solution and place it in the remaining hole on either side of the plate.
- Once all components are in the KitAlysis Benchtop Inertion Box, close the lid and purge for an additional 5 minutes. Leave nitrogen flowing for remainder of set up.
- Purge needle and syringe in a nitrogen diffuser hole 2x by pulling and then pushing plunger. Using purged needle and syringe, add required solvent amounts to open substrate mixture vials. DO NOT add base solution to any substrate mixture-it is added directly to each of the 24 reaction vials as the last step.
- Stir mixtures until in solution (1-2 min). For slurries, see “additional tips” below.
- While mixtures are stirring, carefully remove the cap mat from the 24-vial, preloaded catalysts in the reaction block.
- Dose stock solutions (using a new pipette tip each time)
- Dose 100 µL of “DMAc Substrate Mixture A” to vials A1-A6 according to scheme below.
- Dose 100 µL “Toluene Substrate Mixture B” into vials B1-B6 according to scheme below.
- Dose 100 µL “n-butanol Substrate Mixture C”, to vials C1-C6 according to scheme below.
- Dose 100 µL and “THF Substrate Mixture D” into vials D1-D6 according to scheme below.
You may have a very small amount of excess solution remaining for each mixture. Save it as a reaction standard for HPLC/TLC later.
- Dose 20 µL of the aq. 1.5 M K3PO4 solution mixtures to all vials A1-D6. It is helpful to say the vial location to yourself as you go along to keep track (A1, A2, etc).

Figure 1.Dose instructions for DMAc, Toluene, n-butanol, THF, and K3PO4
- After all substrate mixtures and base have been dosed according to recipe, take the KitAlysis 24-Well Reaction Block lid and line up the screws with the holes in the plate. Ensure the temperature probe holes line up on both the lid and the block.
- Screw on lid according to directions and pattern shown in the “additional tips” section below. Before removing KitAlysis 24-Well Reaction Block from the Inertion Box, ensure that the lid is evenly sealed onto the base. Do this visual check as you go along to avoid having to unscrew the lid and screw again.
- Once completely sealed, remove the KitAlysis 24-Well Reaction Block from the Inertion Box. Place on preheated hot plate with probe through the lid and inserted into the block. Heat at 60 °C overnight stirring at or near 300 rpm. Higher or lower temperatures may be optimal, but 60 °C is a good starting point.
- Make the quench solution/internal standard in a bottle with a replaceable lid following the recipe below for HPLC analysis. TLC is also possible.
- At reaction completion, follow the below Work-Up Procedure to quench reactions, make them more suitable for analysis, and add internal standard.
Quench Solution/Internal Standard Recipe
- 49 mL CH3CN
- 1 mL AcOH
- 15.4 mg Biphenyl (KitAlysis Internal Standard provided in the kit) There is excess in the bottle so be sure to weigh it out.
Note: This recipe makes 50 mL which is enough stock solution for all four screening sets in the KitAlysis Suzuki-Miyaura Cross-Coupling Reaction Screening Kit. The amount of internal standard is 10 mol% per reaction. So a big product peak to small internal standard indicates a good reaction. Integrate to compare reactions against one another (product/internal standard).
Work-up Procedure and Analysis
- Cool Reaction Block. Remove lid using small, non-torque KitAlysis screwdriver.
- Check each vial for solvent loss, and record.
- Aliquot 500 µL of prepared quench solution to each vial using a pipette for accuracy.
- Replace lid, tighten middle screw and stir on stir plate (NO HEAT) for 2-3 minutes. DO NOT INVERT BLOCK.
- After 2-3 minutes of stirring, let plate rest (without stirring) for 5 minutes to allow insoluble material to settle out of solution to the bottom of the vials.
- While plate is resting, add 700 µL of acetonitrile to each 24 individually labeled (A1, A2 etc,) HPLC vials or to each of 24 wells of a 96-well HPLC/UPLC auto sampler block (see “additional recommended materials” below for suggestions on the auto sampler block and cap mat.
- Remove lid on KitAlysis 24-Well Reaction Block carefully.
- Using a clean pipette each time, remove a 25 µL aliquot from each vial into corresponding HPLC vials or HPLC block . Be careful to pull material from the top of the vials to avoid any precipitate.
- Run on HPLC auto sampler. You may need to adjust the amount of acetonitrile from the suggested 700 µL to accommodate your unique HPLC system.
Additional Tips
Sealing the Plate-screwing down the cover
Sealing the plate properly is critical to success. The key is light, even pressure to the lid of the block to keep the cover flat while sealing.
- Line-up screws making sure that the base and lid temperature probe holes line-up in the KitAlysis 24-Well Reaction Block.
- Initially flush: Using your thumb and forefinger, press the lid of the box until it becomes flush with the vials. Then, using the KitAlysis Torque Screwdriver, insert screws until flush, but not tight, with the top of the box, following the cross pattern provided below. Check to see that the lid is evenly sealed onto the base on all sides. Do this visual check to avoid having to unscrew the lid and screw again.
- Tighten: Repeat the same pattern until the KitAlysis Torque Screwdriver “clicks” indicating complete tightness. Go around the block once more for a final check to ensure all screws are tight.
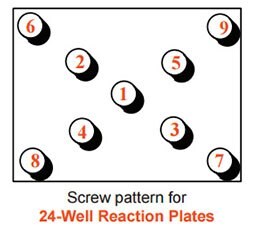
Figure 2.Screw pattern for 24-well reaction plates
Probe for Hot Plate Does Not Fit into Reaction Block Hole
Use a small oil bath or other metal block (for best results) in the back of the hot plate and place the probe in there. Place reaction block as close to the center of the hot plate as possible for more even stirring.
Slurry Additions
Due to narrow opening of pipette tips, slurry additions will result in blockages. To aid in uniform dosing, simply snip off the end of the tip at the first marker (~10 mm). To ensure an evenly dispersed aliquot, it is critical that the mixture is stirring well while you draw aliquots for dosing into the corresponding reaction vial.

Figure 3.Snip approx. 10 mm from end of pipette
To continue reading please sign in or create an account.
Don't Have An Account?