Select a Size
About This Item
Skip To
Agency
ChP
JP
NF
Ph. Eur.
Quality Level
product line
EMPROVE® EXPERT
form
solid
feature
RNase and DNase free
impurities
≤0.3 IU/g endotoxin (low)
pH
7 (20 °C, 100 g/L in H2O)
mp
185-187 °C (lit.)
application(s)
liquid formulation
parenterals
pharma/biopharma processes
pharmaceutical
shipped in
ambient
storage temp.
2-30°C
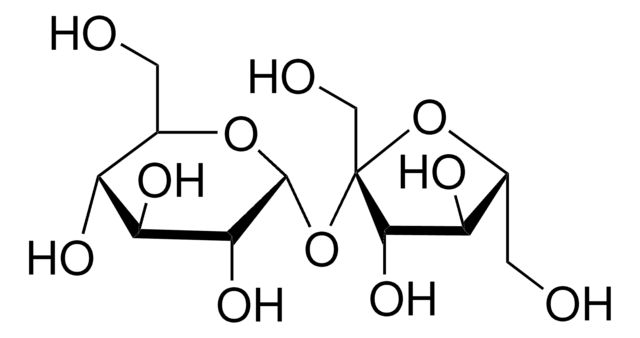
SMILES string
OC[C@H]1O[C@H](O[C@]2(CO)O[C@H](CO)[C@@H](O)[C@@H]2O)[C@H](O)[C@@H](O)[C@@H]1O
InChI
1S/C12H22O11/c13-1-4-6(16)8(18)9(19)11(21-4)23-12(3-15)10(20)7(17)5(2-14)22-12/h4-11,13-20H,1-3H2/t4-,5-,6-,7-,8+,9-,10+,11-,12+/m1/s1
InChI key
CZMRCDWAGMRECN-UGDNZRGBSA-N
Looking for similar products? Visit Product Comparison Guide
1 of 4
This Item | 1.07653 | S1600000 | PHR1001 |
|---|---|---|---|
| application(s) liquid formulation | application(s) liquid formulation | application(s) pharmaceutical (small molecule) | application(s) pharmaceutical (small molecule) |
| agency ChP, NF, JP, Ph. Eur. | agency BP, ChP, JP, NF, Ph. Eur. | agency - | agency traceable to BP 818, traceable to Ph. Eur. S1600000, traceable to USP 1623637 |
| form solid | form solid | form - | form - |
| product line EMPROVE® EXPERT | product line EMPROVE® ESSENTIAL | product line - | product line - |
| feature RNase and DNase free | feature - | feature - | feature - |
| impurities ≤0.3 IU/g endotoxin (low) | impurities - | impurities - | impurities - |
General description
As part of our Emprove® Program, our raw materials are offered with extensive documentation facilitating compliance of your pharma and biopharma product, full supply chain transparency and risk mitigation. Our SAFC® portfolio of high-quality products for biopharmaceutical and pharmaceutical formulation and production withstands strict quality control procedures and is produced according to applicable cGMP guidelines.
Application
We have developed a new filtration-based manufacturing process leading to a sucrose grade with unique quality characteristics: Sucrose Emprove® Expert Ph Eur, ChP, JP, NF is low in endotoxins and has extensively reduced levels of nanoparticulate impurities which can negatively influence the stability of biomolecule formulations, making it the ideal choice for your high-risk applications.
Legal Information
related product
used together
Storage Class Code
11 - Combustible Solids
WGK
WGK 1
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Stay compliant with GMP using the Emprove® Program
Accelerate qualification, maintain compliance, and save time with Emprove® Dossiers.
Note: you will be brought to EmproveSuite.com. Your username and password are separate from SigmaAldrich.com and may differ.
Register for free and access Emprove® Dossiers within minutes. Upgrade your subscription plan to unlock advanced and customized content in the Emprove® Suite
Not finding what you are looking for? Visit the Emprove® Program Help page.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Active Filters
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service