Dissolution Testing Filtration
- Why is Filtration Important in Dissolution Testing?
- What is the Primary Difference Between Frits and Membrane Filters?
- How is Membrane Pore Size Measured?
- How are Membrane Filters Selected for Dissolution Testing?
- Why are Hydrophilic PTFE Membranes Typically Used in Dissolution Testing?
- How is Automation Used in Dissolution Testing?
- Non-Sterile Millex® Dissolution Testing Compatibility
Related Filtration Products for Dissolution Testing
- 33MM Millex® Syringe Filters
- Automation Compatible Millex® Syringe Filters with Domed Housing
- Automation Compatible Millex® Syringe Filter Compatibility by System Manufacturer
Critical in new formulation development and quality control testing during manufacturing, dissolution testing is used to measure active pharmaceutical ingredient (API) release over time. Dissolution testing is required for all solid, oral formulations and utilizes filtration to stop the dissolution process. Depending on the membrane type and format selected, the dissolution testing method and results may be impacted.
Why is filtration important in dissolution testing?
Samples pulled from dissolution baths during testing contain both dissolved and undissolved API. Microporous membranes, such as those used in Millex® syringe filters, separate undissolved from dissolved API, stopping the dissolution process. In addition to capturing API release and stability at the given time point, filtration removes particulates to prepare the sample for downstream testing methods, including HPLC. Samples for dissolution testing are typically filtered using either fritted or membrane filters.
What is the primary difference between frits and membrane filters?
Fritted filters and membrane filters differ by two main criteria: pore size and pore size distribution. Membrane pore sizes in a syringe filter (or membrane filter in a filter holder) are usually between 0.2-1 μm, whereas the pore size of fritted filters is generally between 5–70 μm. Larger pores in fritted filters may allow undissolved API particles to pass through, impacting the quantitation of API and downstream HPLC testing.
Pore size is determined differently for membrane and fritted filters, which leads to differences in their pore size distribution. The pore size reported for membrane filters refers to the largest pore in a membrane filter, while the pore size of fritted filters refers to the average pore size. During filtration, membrane filters retain all particles larger than the pore size listed, while fritted filters may not be able to retain all particles that are larger than the frit pore size, depending on what the actual size is of the largest pore. As the largest pore size of a fritted filter is not reported, the only way to ensure all particles above a certain size threshold are retained is to use a membrane filter.
How is membrane pore size measured?
Due to their small size, the pore size of microporous membranes is typically determined by a bubble point measurement. During this test, the membrane is wetted, either with water or an organic solvent, and placed in a holder, where the wetting liquid is on one side of the membrane and air is on the other. Air is slowly pressurized through the membrane and at a certain pressure the liquid in the membrane pores is displaced, creating a steady stream of bubbles on the liquid side. The pressure at which this displacement occurs is called the bubble point. Bubble point is inversely proportional to membrane pore size, and thus this relationship is used to determine the largest pore size in the membrane filter.
For sterilizing membranes, pore size is also determined by bacterial (0.22 μm) or mycoplasma (0.1 μm) challenge.
How are membrane filters selected for dissolution testing?
There are three key parameters in membrane filter selection for dissolution testing:
- Chemical compatibility
- Low levels of extractables
- Low analyte or API binding
To avoid membrane failure during filtration, the membrane material must be chemically compatible with the solution being filtered. Verify the compatibility of all our Millipore® membranes with our chemical compatibility tables. The presence of extractables in the filtrate or the loss of analytes due to non-specific binding to the membrane, will alter quantitative results obtained in downstream analysis.
Why are hydrophilic PTFE membranes typically used in dissolution testing?
PTFE, or polytetrafluoroethylene, membranes have very broad chemical compatibility, making them ideal for use with a variety of solutions. While native PTFE is hydrophobic, we offer surface-treated, hydrophilic PTFE membranes that allow users to filter aqueous solutions while retaining high chemical compatibility. Our hydrophilic PTFE membranes undergo additional washing steps during membrane coating, resulting in membranes with very low extractable levels. These low extractable levels ensure the filtration device does not add contaminants into the filtrate, which can complicate the downstream HPLC analysis and analyte quantitation. Hydrophilic PTFE membranes also exhibit very low analyte binding, ensuring analyte quantitation, including APIs or other compounds present in the formulation, is not negatively impacted by the analyte binding to the membrane. With the three selection criteria in mind, hydrophilic PTFE membranes offer a universal solution for filtration in dissolution testing.
How is automation used in dissolution testing?
With the rapid pace of the pharmaceutical industry, sample preparation in drug screening and development is often automated. While this advance in technology saves time and resources, standard syringe filters often fail in automated systems due to high back pressure, filter misalignment, or filter jams due to shingling (Figure 1).
Non-Sterile Millex® Dissolution Testing Compatibility
Low drug-binding and low extractable non-sterile Millex® syringe filters are compatible with numerous dissolution and other automated sample preparation stations. Automated samplers sold by individual dissolution instrumentation manufacturers will have varying requirements and specifications that necessitate different styles, shapes, fittings, and sizes of syringe filter. This chart will instruct in selecting compatible syringe filters for your specific equipment according to make, model, membrane type, pore size, filter diameter, and pack size.
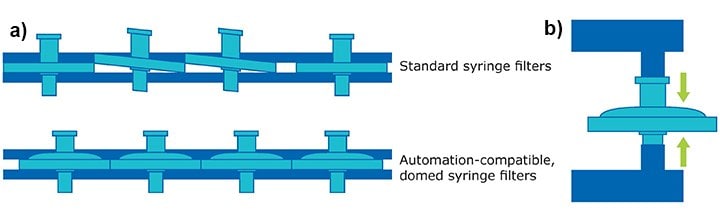
Figure 1.
a) The convex, domed housing of our automation-compatible syringe filters prevent shingling in automated systems compared to standard syringe filters.
b) The pressure resistant housing prevents bursting and the Luer-Lok® connection provides precise alignment within automated systems.
Our automation-compatible syringe filters alleviate these issues with:
- Domed housing to prevent shingling (Figure 1a)
- Pressure-resistant housing to prevent bursting (Figure 1b)
- Luer-Lok® connection optimized for precise alignment and fit
Alternatives to manual and automated filtration
While automation can be useful in multisample preparation, the high cost of the required infrastructure limits its application to high-volume industrial applications. Our vacuum-driven Samplicity® filtration device offers a solution for scientists that need to filter more than eight samples daily, but do not meet the threshold for automated sample preparation. Using Millex® Syringe Filters with adaptor funnels, researchers can filter up to eight samples with simultaneously into standard 2 mL HPLC sample vials.
| Membrane Type | Pore Size (µm) | Diameter | Product No. |
|---|---|---|---|
| Glass fiber | 1.0 | 25mm | SLPBDZ5 |
| PVDF | 0.45 | 25mm | SLHVDZ5NZ |
| PVDF with glass fiber prefilter | 0.45 | 25mm | SLHVBZ5 |
| Hydrophilic PTFE | 0.45 | 25mm | SLCRDZ5 |
| 0.20 | 25mm | SLGNDZ5 | |
| Hydrophilic PTFE with glass fiber prefilter | 0.45 | 25mm | SLCRBZ5 |
| Nylon | 0.45 | 25mm | SLHNDZ5 |
| Nylon with glass fiber prefilter | 0.45 | 25mm | SLHNBZ5NK |
| 0.45 | 25mm | SLHNBZ5 | |
| Hydrophilic PTFE | 0.20 | 25mm | SLLGDZ5 |
Sotax
PharmaTest
Logan
| Sampler Model | Membrane Type | Diameter | Product No. |
|---|---|---|---|
| FTC-1200: Automated Filter Changer | Glass fiber | 25mm | SLPBDZ5 |
| Durapore® PVDF membrane | 25mm | SLHVDZ5 | |
| PVDF with glass fiber pre-filter | 25mm | SLHVBZ5 | |
| Nylon | 25mm | SLGNDZ5 | |
| Nylon | 25mm | SLHNDZ5 | |
| Nylon with glass fiber pre-filter | 25mm | SLHNBZ5NK | |
| Philic PTFE | 25mm | SLLGDZ5 | |
| Philic PTFE | 25mm | SLCRDZ5 | |
| LCR with glass fiber pre-filter | 25mm | SLCRBZ5 | |
| Durapore® PVDF membrane | 25mm | SLHVDZ5NZ | |
| HPF/HV | 25mm | SLHVMZ5NZ | |
| HPF/Nylon | 25mm | SLHNMZ5NZ | |
| HPF/Nylon | 25mm | SLGNM25NS | |
| HPF/LG/LCR | 25mm | SLLGM25NS | |
| HPF/LG/LCR | 25mm | SLCRM25NK | |
| Nylon | 25mm | SLHNM25 | |
| Durapore® PVDF membrane | 25mm | SLHVM25 | |
| HPF/LG/LCR | 25mm | SLCRM25NS |
To continue reading please sign in or create an account.
Don't Have An Account?