Surface & Personnel Testing
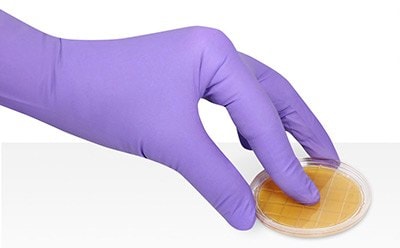
Surface and personnel testing are methods to collect, detect, and characterize the levels of various microorganisms to monitor the efficiency of disinfection routines in critical and non-critical manufacturing areas. Hygiene standards in a production environment are directly linked to the microbiological safety and quality of the finished products. To protect consumers, several government agencies have developed methods, including EU cGMP, U.S. FDA Aseptic Guidance, and USP <1116>, which apply to regulate industries like pharmaceutical, cosmetics, food, and beverages.
Featured Categories
Explore versatile air samplers based on various principles for tailored technology solutions. Designed for food, cleanrooms, hazardous areas, and more.
Hygiene monitoring verifies the effectiveness of your sanitization routine using methods like ATP testing, NAD testing, protein testing or swab sampling for food and beverage HACCP and cannabis microbiology testing.
Traditional Surface Monitoring
To ensure the safety of a product, it is key to monitor the cleanliness of surfaces in the production area, such as tables, walls, or doors, and in isolators and cleanrooms. These surfaces and areas must be free of microorganisms to reduce the risk of cross-contamination, which could make the product unsafe for use or consumption.
Aseptic and controlled environments require frequent monitoring by surface sampling methods. Flat surfaces can be sampled by pressing contact plates or slides on the surface with uniform and steady pressure for a few seconds. The sample plates or slides are then incubated for the microorganisms to grow, before enumerating the grown bacteria cultures. The incubation conditions differ depending on microflora composition and regulatory requirements. Curved surfaces and hard-to-access areas can be monitored by the swab test method.
Rapid Surface Monitoring
Manufacturers who require fast results can use rapid methods to quantify microbial residue post-sanitation and pre-operation. Such methods include colorimetric based assays requiring no instrument or sampling swabs requiring an instrument reader such as a luminometer. The need for rapid quality control methods is especially high in the food and beverages industry. Due to the perishable nature of their manufactured goods, but also in other industries to keep up with increasing production demand.
Personnel Monitoring
Personnel monitoring is performed to ensure the maintenance of the sterile environment. The majority of contamination within a facility can be traced to humans working in cleanrooms. Specific procedures for gowning, and sterilizing, need to be followed by the personnel before entering and exiting the aseptic cleanroom.
Visit our document search for data sheets, certificates and technical documentation.
Related Articles
- Contact Plates and Swabs for Personnel and Surface Monitoring in Cleanrooms and Isolators
- The ICR-Swab is a swab system for environmental monitoring of dry surfaces, such as filling needles, equipment recesses, uneven surfaces and tubing in clean rooms and isolators.
- The ICR swabs and ICR contact plates are designed for environmental monitoring i.e., detection and enumeration of microbial contamination on dry, flat, and hard-to-access surfaces in cleanrooms and isolators.
- Halal-certified culture media prevent non-halal ingredient contamination, enabling microbiological QC testing without losing certification.
- Discover the importance of data integrity in environmental monitoring. Learn how digital management systems can enhance compliance, safety, and efficiency in data handling.
- See All (4)
Find More Articles and Protocols
How Can We Help
In case of any questions, please submit a customer support request
or talk to our customer service team:
Email [email protected]
or call +1 (800) 244-1173
Additional Support
- Calculators & Apps
Web Toolbox - science research tools and resources for analytical chemistry, life science, chemical synthesis and materials science.
- Customer Support Request
Customer support including help with orders, products, accounts, and website technical issues.
- FAQ
Explore our Frequently Asked Questions for answers to commonly asked questions about our products and services.
To continue reading please sign in or create an account.
Don't Have An Account?













