Imprint ® Methylated DNA Quantification Kit (MDQ1) Protocol
- Product Description
- Kit Components
- Storage/Stability
- Workflow
- Reagents Preparation
- Procedure
- Calculations
- Troubleshooting
- Materials and Reagents Required but Not Provided
- Materials
- References
Note: Storage Temperature - see Storage/Stability section for individual component storage conditions
Product Description
It has been well demonstrated that DNA methylation plays an important role in the regulation of gene expression. At any given time about 70% of all CpG dinucleotides in the mammalian genome are methylated.1 Hypo- and hyper-methylation across the genome contribute to changes in gene expression. Determining the methylation status of DNA is an important tool in epigenetic studies.8,11
Shifts away from normal global DNA methylation levels have been observed in various cancers, neurological disorders, autoimmune diseases (such as lupus and cardiovascular diseases), and aging.10
The Imprint Methylated DNA Quantification Kit provides a high-throughput, non-radioactive means of measuring global DNA methylation shifts in about half of a day.
This kit contains all reagents required to detect relative levels of methylated DNA. Up to 200 ng of purified DNA is bound to the wells of the assay strip. The methylated DNA is detected using the Capture and Detection antibodies, then quantified colorimetrically. The amount of methylated DNA present in the sample is proportional to the absorbance measured.
A methylated DNA control is provided. A standard curve can be performed (range: 10-100 ng) or a single quantity of DNA can be used as a positive control. Because global methylation can vary from tissue to tissue, and from normal and diseased states, it is advised to run replicate samples at different DNA concentrations to ensure that the signal generated falls within the detection range of the plate reader used.
The kit will allow the user to determine relative methylation states of two different DNA samples.
Kit Components | |||
|---|---|---|---|
| Reagent | Catalog Number | 48 Rxn | 96 Rxn |
| 10x Wash Buffer | W4267 | 11 mL | 22 mL |
| DNA Binding Solution | D8568 | 1.5 mL | 3 mL |
| Methylated Control DNA (50 ng/μL) | M8570 | 16 μL | 32 μL |
| Block Solution | B6561 | 1.5 mL | 20 mL |
| Capture Antibody | C3493 | 10 μL | 20 μL |
| Detection Antibody | D8818 | 10 μL | 25 μL |
| Developing Solution | D8693 | 6 mL | 12 mL |
| Stop Solution | S3447 | 3 mL | 6 mL |
| 8 well Assay Strips | A4231 | 6 strips | 12 strips |
| Reagent | Catalog Number | Storage Temperature |
|---|---|---|
| 10x Wash Buffer | W4267 | 2-8 °C |
| DNA Binding Solution | D8568 | 2-8 °C |
| Methylated Control DNA (50 ng/μL) | M8570 | -20 °C |
| Block Solution | B6561 | -20 °C |
| Capture Antibody | C3493 | 2-8 °C |
| Detection Antibody | D8818 | 2-8 °C |
| Developing Solution | D8693 | 2-8 °C |
| Stop Solution | S3447 | 2-8 °C |
| 8 well Assay Strips | A4231 | 2-8 °C |
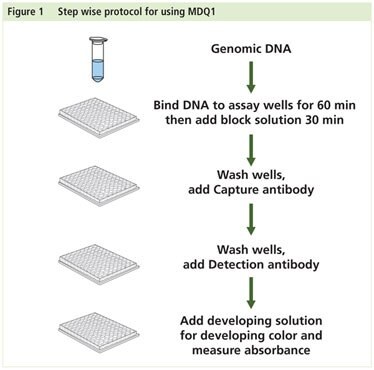
Preparation Instructions
1 x Wash Buffer
For the 48-reaction kit, add 11 mL of 10x Wash Buffer to 99 mL of Water, Molecular Biology Reagent. Store at room temperature for up to six months.
For the 96-reaction kit, add 22 mL of 10x Wash Buffer to 198 mL of Water, Molecular Biology Reagent. Store at room temperature for up to six months.
DNA Dilutions
Dilute sample or control DNA in DNA Binding Solution. Each well can bind a maximum of 200 ng of total DNA.
Procedure
DNA Binding
- Add 30 μL of DNA diluted in DNA Binding Solution to each well. Ensure the solution coats the bottom of the well by gently tilting from side to side. The DNA Binding Solution alone can be used as a blank.
- Cover and incubate at 37 °C for 60 minutes.
- Add 150 μL of Block Solution directly to each well.
- Cover and incubate at 37 °C for 30 minutes.
- Remove DNA and Block Solutions from each well.
- Wash three times with 150 μL of 1x Wash Buffer.
Methylated DNA Capture
- Dilute Capture Antibody 1:1000 in 1x Wash Buffer.
- Add 50 μL of diluted Capture Antibody to each well.
- Cover and incubate at room temperature for 60 minutes.
- Remove diluted Capture Antibody from well.
- Wash four times with 150 μL of 1x Wash Buffer.
- Dilute Detection Antibody 1:1000 in 1x Wash Buffer.
- Add 50 μL of diluted Detection Antibody to each well.
- Cover and incubate at room temperature for 30 minutes.
- Remove diluted Detection Antibody from well.
- Wash five times with 150 μL of 1x Wash Buffer.
Detection
- Add 100 μL of Developing Solution to each well.
- Cover and incubate at room temperature away from light for 1-10 minutes. Monitor reaction for color change. The solution will turn blue.
- Add 50 μL of Stop Solution to each well. The reaction will turn yellow.
- Read the absorbance at 450 nm on a plate reader.
- Calculate relative global methylation levels.
Calculations
To calculate methylation levels relative to the Methylated Control DNA, use the following steps.
Single Point Method
Percent methylation relative to the Methylated Control DNA
- Average the A450 replicates for the blank, sample(s) and Methylated Control DNA
- Perform the following calculation to obtain the percent methylation of the sample(s) relative to the Methylated Control DNA
[(A450 av Sample - A450 av Blank)/(A450 av Methylated Control DNA - A450 av Blank)] x 100
- Sample calculation:
Average A450 Blank = 0.090
Average A450 for 60 ng Methylated Control DNA = 0.785
Average A450 for 60 ng sample DNA = 0.654
[(0.654 – 0.090) / (0.785 – 0.090] x 100 = 81.15%
The sample DNA has a global methylation level that is 81.15% relative to the Methylated Control DNA
Standard Curve Method
ng Methylated DNA in sample(s) from standard curve of Methylated Control DNA
- Create a standard curve by plotting
(A450 av Methylated Control DNA - A450 av Blank) versus ng of the Methylated Control DNA.
See sample curve.
- Calculate the slope of the standard curve using linear regression
- For each value of (A450 av Sample - A450 av Blank), calculate the ng methylated DNA as below:
ng methylated DNA in sample = [(A450 av Sample - A450 av Blank) – intercept] / slope
Note: in many, or most cases, the intercept will be zero.
Troubleshooting | ||
|---|---|---|
| Observation | Cause | Recommended Solution |
| High Background Present in Blank (>0.2 OD) | Inappropriate/inconsistent washing | Perform all wash steps as indicated in the protocol. If necessary, one wash can be added at each of the wash steps. |
| Contaminating DNA | Ensure a clean tip is used when applying DNA Binding Solution. When handling plate, ensure that no splashing occurs. | |
| Over-development | Monitor development of blue color once the Developing Solution has been added. Do not allow the reaction to incubate more than ten minutes. | |
| No Signal for DNA-Containing Samples (>0.2 OD) | Addition of Reagents | Ensure reagents are added in the appropriate order and none of the procedure steps have been omitted. |
| Washing | Ensure the wells of the assay strip are not washed prior to the application of the DNA sample. | |
| Incubation time and temperatures | Ensure incubation time and temperatures correspond to the written protocol. | |
| Addition of DNA to well | Ensure DNA is mixed thoroughly prior to application. The wells of the assay strip bind up to 200 ng of total DNA. The methylated control DNA (Catalog Number M8570) included in the kit can be detected at the 10 ng level. | |
| Degradation of DNA | Ensure DNA is stored at the appropriate temperature. | |
Materials
Response not successful: Received status code 500
References
Imprint is a registered trademark of Sigma-Aldrich Co. LLC.
To continue reading please sign in or create an account.
Don't Have An Account?