Tips & Tricks for Thin-Layer Chromatography
Monika Bäumle, Global Product Manager TLC, Ilona Matus, Analytical Sciences Liaison
MilliporeSigma
Introduction
Thin-layer chromatography (TLC) is a fast, easy-to- use, and highly versatile separation technique for qualitative and quantitative analysis. It is ideal for rapid identification, screening, and reaction monitoring. Its high matrix tolerance and the possibility to separate many samples in parallel makes TLC highly time- & cost-efficient.
Working Principle of TLC
The general thin-layer chromatography process is simple, however it includes a range of steps and some precautions need to be considered (Figure 1).
One example is the gas phase in the development chamber, which influences the TLC process. Therefore, it is critical to maintain controlled gas and humidity conditions to obtain reproducible performance and accurate TLC results. In the following we address some of the important aspects, which should be considered at different steps.
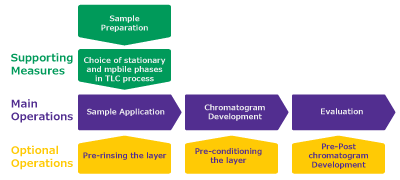
Figure 1.TLC process.
Storage and Handling of TLC Plates
TLC layers are highly active materials and can adsorb moisture and contaminants originating from the lab environment. Therefore, a storage of plates in a clean and dry environment is recommended (e.g. in a desiccator). If possible, wrap the plates in aluminum foil and keep them away from chemical fumes and vapor.
Preparation of TLC plate: Pre-Rinsing (Washing) and Activation of the Layer
Contaminants and moisture from the environment can alter the plate’s performance, especially when not stored properly after opening a package. In addition, plates may have impurities from binders, packaging, or previous handling. These contaminants can/should be removed by pre-rinsing the layer. This can be performed by either dipping them in a solvent (once or twice, 1-7 minutes) or by a blank run of the TLC plate e.g., with methanol. Mind the chromatographic direction, because impurities will concentrate at the top edge of the plate.
To remove bound water at the polar functional groups of the phase, it is advised to heat the plate for 20 – 30 min at 120 °C (in a clean oven) for a proper plate activation.
TLC Sample Preparation
Thin-Layer Chromatography plates are single-use devices, hence they do not bear the risk for cross-contamination into future analysis. Because of this, sample preparation can typically be simplified and is therefore less time-consuming compared to HPLC. A sample crushing/homogenization and extraction with an appropriate diluent is recommended (e.g. for solid samples), eventually followed by filtration and concentration steps.
Choice of Stationary Phase in TLC
The choice of the TLC stationary phase is crucial as it determines the selectivity and separation performance. There is a broad range of options available – based on silica, aluminum oxide or cellulose, modified or non- modified. The selection should be in relation to the properties of your sample and your application goals. TLC can be run in two modes, normal and reversed phase mode. In normal phase (NP) mode, the mobile phase is less polar than the stationary phase on the TLC plate, whereas in reversed phase (RP), the mobile phase is more polar than the stationary phase. More than 80% of all TLC separations are done on silica gel as stationary phase, either bare or modified with e.g. C18.
How to Choose Solvent System for TLC?
The choice of the mobile phase in TLC is another critical factor for an efficient separation result. The solvent dissolves the sample components on the sorbent layer and moves them across the plate. Ideal mobile phases transport all components from the baseline having final RF (retention factor) values between 0.15 up to 0.85 (ideally 0.2 – 0.6). The retention factor is defined as the distance traveled by the substance divided by the distance traveled by the solvent. Usually, these are also described as hRF, which is defined as 100x RF. When a method is developed from scratch, typically a mixture of a polar and non-polar solvent is used as a starting point. To increase RF values in NP-TLC, an increase in the polarity of the mobile phase is needed. In case, a reduction of RF is required, a decrease in polarity needs to be done. A very common mobile phase system in NP-TLC contains hexane and 10 – 50% ethyl acetate (EtOAc). Other prominent solvent systems are based on methanol and dichloromethane, toluene or acetone. In RP-TLC, the solvent systems are commonly mixtures based on water, methanol, acetonitrile, or aqueous buffers.
The employed solvents and solvent blends should have an adequate purity and stability, low viscosity, low vapor pressure, and low toxicity, if possible.
The addition of certain modifiers (basic or acidic) might improve separation results.
A detailed systematic approach to find the ideal solvent systems can be followed based on the published data.1
Sample Application
Samples can be applied as spots or bands by contact or spraying. Sample application by spraying in a band enables improved separation result and is the preferred method when having larger sample volume. There is a special equipment available to spray sample solutions onto a plate. This method avoids direct contact with the TLC layer and is typically used for band application.
The polarity of the sample diluent is a factor to be considered. In NP-TLC, non-polar diluents are used, like n-hexane, which ensure that the substances remain at the application point. However, with more polar diluents (e.g. toluene, dichloromethane, methanol), the sample substances are transported toward the edge of the “wet zone” (spot at the starting line), and typically form a circular chromatographic spot at the starting area (see top of Figure 2). Like in chromatographic development, peaks with an almost Gaussian distribution are achieved, which is widening with increasing polarity of the diluent (bottom Figure 2).
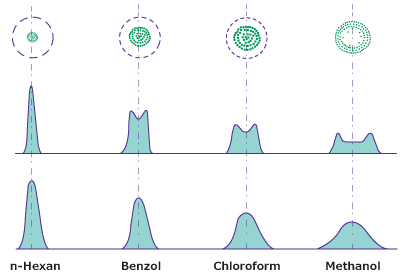
Figure 2.Substance distribution in a sample spot after application on a TLC plate as a function of the solvent.
Based on the number of samples being analyzed, select a suitable plate size or cut a larger plate to the needed dimensions. Mark the application zone with a pencil by drawing lines across the plate. Take care not to damage/scratch the layer surface as this can influence the flow behavior and lead to errors. Be careful not to apply the sample too close to the bottom edge of the plate (8 mm from lower edge), as this can cause the starting point to spread/leach into the mobile phase. The sample volume to apply depends on the goal of the analysis and the concentration of the sample solutions. Typically, a sample volume of 0.5-2.0 µL is recommended for identity tests, and a maximum of 10 µL for purity testing. The higher the sample volume, the more volatile and non-polar (for NP) the diluent should be and the slower the application should be carried out.
Drying Plates Prior to Development
After the sample is applied on the plate, the sample diluent must be completely removed by careful drying the plates prior to development. Avoid contamination with fumes during this step and chose an appropriate temperature in order not to cause sample diffusion or loss. The temperature depends on the kind of substances, their stability and boiling points.
Pre-Conditioning the Layer/ Humidity Control
Unless special precautions are taken during sample application, humidity in the laboratory can diminish the activity of the TLC layer within minutes, as an equilibrium is quickly established between the lab atmosphere and the sorbent. Pre-conditioning the TLC plate before development helps to avoid its deterioration. Therefore, condition your plates (after sample application) for 45 min over a saturated salt solution in a closed chamber. Depending on the relative humidity desired, several saturated salt solutions are typically recommended, for example a relative humidity of 33%, can be achieved by using a saturated MgCl2 solution. Ensure, that there is still undissolved salt in the solution to ensure saturation. After conditioning it is important to develop the plate immediately to prevent recurrence of alterations.
Chromatogram/Plate Development
The development of the TLC plates can be done via various techniques, e.g. one-dimensional, two- dimensional or via forced flow processes. One-dimensional can be single or multiple development, and is run vertically, horizontally, or circular. In most cases, the separation is done vertically in a development chamber.
First add the freshly prepared mobile phase in the development chamber to a level of max. 0.5 cm (immersion line). Before starting the development process, an equilibrium between the liquid mobile phase and the gas phase is required to achieve a chamber saturation. To facilitate that faster, add a saturating pad or place a filter paper inside the chamber (chamber wall) and equilibrate for 20 min (keep the chamber closed). After chamber saturation, place the plate quickly inside the chamber. As soon as the mobile phase has travelled two-thirds of the plate dimension (max. up to 1 cm from the top), remove the plate and mark the solvent front. After development, the plate should be properly dried to remove residual solvents before visualization.
Derivatization in TLC
After separation, typically a derivatization is done to enable visualization of analytes and enhancement of detection, if the sample components are colorless, or do not fluoresce. The ideally suited derivatization/ detection reagent is dependent on the target analytes and desired detection method. It may be applied before development (pre-chromatographic derivatization), with the solvent system (in-situ derivatization), or after development (post-chromatographic derivatization).
For post-chromatographic derivatization or visualization of the analytes, the reagent is applied to the plate by spraying or dipping. The advantages of spraying are its high flexibility and low reagent amount required. However, manual spraying often does not provide sufficient reproducibility.
Derivatization by dipping allows for more homogeneous and reproducible derivatization but requires more reagent. Besides that, background coloration may occur. Make sure to carefully wipe the back site of the plate after derivatization and before reading out.
How to read TLC plates?
Performing TLC in a proper way will provide you reproducible and precise results both for qualitative and quantitative analysis.
We will cover more Tips and Tricks for the visualization and documentation process in a separate article.
Products
Response not successful: Received status code 500
References
To continue reading please sign in or create an account.
Don't Have An Account?