An Introduction to A-Phos, its Applications and Advantages
Introduction
The Suzuki-Miyaura cross-coupling reaction of heteroaryl halides is of interest to the pharmaceutical industry because many biologically active compounds are accessed through this methodology. A need exists for efficient coupling of boronic acids with five-membered heteroaryl halides or six-membered aryl chlorides bearing heteroatom substituents. With many of these types of substrates, catalyst deactivation is noticed, thus, high catalyst loading is typically required to achieve good yields. This is an efficient new process to access biaryls substituted with heteroatoms.
Advantages
- Air-stable catalyst
- Reaction done in aqueous solution
- Low catalysis loading
- Access to biaryls substituted with heteroatoms
Representative Applications
Guram and coworkers, from Amgen, reported the synthesis and the activities of a set of air-stable palladium catalysts. These catalysts are based on bulky dialkylphenylphosphines where the phenyl is substituted at the para position with an amine, an alcohol or trifluoromethyl groups. Utilizing these palladium complexes, Guram et al. evaluated their efficiency for the coupling reaction of heteroatom containing substrates. Excellent yields were obtained with 1 mol% loading of the palladium complex for the coupling of a variety of aryl chloride with aryl boronic acids.
In a typical reaction, 1 mol% of the catalyst is loaded with potassium carbonate and refluxed in aqueous mixture for 12 hours. A variety of pyridine derivative were successfully coupled with bulky, electron donating and electron deficient boronic acids with yields up to 99%.
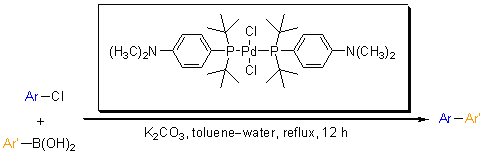
| Product | Yield (%) |
 | 93 |
 | 92 |
 | 93 |
 | 98 |
 | 95 |
 | 94 |
 | 99 |
 | 98 |
| 97 | |
| 96 | |
 | 99 |
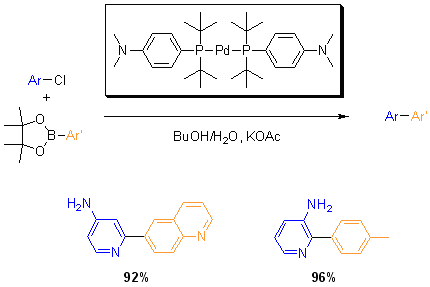
More recently, Guram and co-workers reported the Suzuki coupling of various aryl chloride with aryl boronate esters, offering a greater range of coupling reagents. In this reaction, butanol and water are used as solvents with potassium acetate as a base with 1 mol% loading of the catalyst.
Response not successful: Received status code 500
To continue reading please sign in or create an account.
Don't Have An Account?