Bioprinting for Tissue Engineering and Regenerative Medicine
Chi-Chun Pan1,2, Arnaud Bruyas1, Yunzhi Peter Yang3,4
1Departments of Orthopedic Surgery, 2Mechanical Engineering, 3Materials Science and Engineering, 4Bioengineering Stanford University, 300 Pasteur Drive, Stanford, CA 94305
Material Matters, 2016, 11.2
Introduction
In the past two decades, tissue engineering and regenerative medicine have become important interdisciplinary fields that span biology, chemistry, engineering, and medicine.1,2 These new fields promote the healing and restoration of lost function in damaged or diseased tissues and organs by combining scaffolds, cells, and biological signaling molecules to recreate functional biological substitutes and mimic native tissues and functions.3 One objective of tissue engineering and regenerative medicine is the fabrication of viable tissues and organs for transplantation, but with the exceptions of thin skin and avascular cartilage,4 limited success in human patients has been achieved due to the complexity of tissue biology. The traditional tissue engineering approach includes loading cells onto a solid porous biomaterial, called a scaffold, in the presence or absence of growth factors that encourage cells to form the desired tissues with biomimetic complexity.5 However, the desired result is rarely achieved because the three component mixture does not adequately promote formation of a well-defined spatial distribution of cells, growth factors, and biomaterials at the microscale level that is characteristic of a tissue-like structure. Three-dimensional (3D) printing, also known as additive manufacturing (AM), holds great promise to overcome this limitation in tissue engineering. Because it is a layer-bylayer process, 3D printing enables the formation of complex geometries using multiple materials (Figure 1). 3D printing for tissue engineering has evolved into a new technology, called bioprinting, defined as “the use of material transfer processes for patterning and assembling biologically relevant materials, molecules, cells, tissues, and biodegradable biomaterials with a prescribed organization to accomplish one or more biological functions.”6 In particular, bioprinting enables personalizable and precision medicine by engineering anatomically shaped implants with tissue-like complexity using a patient’s own cells. Currently, 3D bioprinting technologies can be classified into two categories: acellular and cellular constructs.7 Acellular bioprinting is used to manufacture the scaffold and biomaterial itself in the absence of cells during the printing process. Acellular bioprinting offers higher accuracy and greater shape complexity than cellular constructs because the fabrication conditions are less restrictive than methods that require maintenance of cell viability. For cellular bioprinting, cells and other biological agents are integrated into the material during manufacturing in order to fabricate living tissue constructs. It is clear that the printing parameters, biomaterials, and properties of the 3D-printed constructs are, therefore, different in each category because of the presence or absence of cells and biological substances. Here we briefly introduce and discuss these two approaches based on the suitable materials for these constructs and the fabrication processes used to manufacture them. We also discuss current limitations, potential solutions, and future directions in bioprinting.
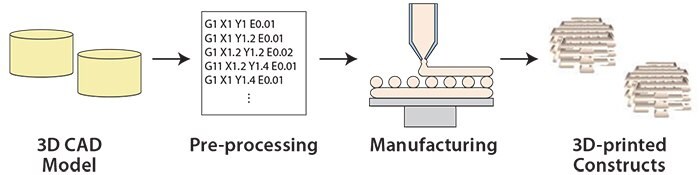
Figure 1.Overview of the 3D printing process.
Manufacturing of Acellular Scaffolds
An acellular scaffold consists of a porous structure that mimics the mechanical and biochemical properties of the extracellular matrix (ECM) and provides mechanical integrity as well as a template for cell attachment in order to stimulate tissue formation.8 Acellular scaffolds must present biocompatible and bioresorbable properties as well as biochemical, biophysical, biomechanical, bioelectrical, and biomagnetic signals.9 Since pores provide room for cell migration and tissue ingrowth, facilitate vasculature formation, and improve cell viability,10 porosity and porous structures are other key features for the scaffold. Thus, the use of AM is highly beneficial, allowing very accurate and repeatable control of the scaffold geometry (and, thus, porosity) while allowing for the potential assembly of tissue-like spatial complexity. A wide range of applications of bioprinted acellular scaffolds have been reported, such as muscular tissues, liver tissues, cartilage, bone, skin, etc.2 The specific material that composes the scaffold and any potential biological agents must be selected to recreate the nature of the engineered tissue. In this section, we focus on constructs with high mechanical strength, typically for engineered bone. The materials and AM processes for acellular scaffolds based on soft engineered tissues (e.g., skin, liver) are similar to the cellladen ones and are, therefore, described in the section “Manufacturing Soft Materials for Cell Encapsulation” later in this article.
Materials
Four categories of materials are highlighted based on their chemical nature. The first category is polymers,11 such as collagen (Product No. C5483, C7624, H4417, etc.), fibrin (Product No. F5386), alginate, chitosan (Product No. 448869, 448877, 419419, etc.), poly(lactic acid) (PLA) (Product No. 764590, 765112, 764698, etc.), poly(glycolic acid) (PGA) (Product No. 457620 and 46746), polycaprolactone (PCL), and poly(propylene fumarate) (PPF). They can be highly bioresorbable and highly flexible in terms of chemical composition and processability. However, polymerbased scaffolds show a rapid decrease in stiffness over time once implanted. Calcium phosphate (CaPs) (Product No. 21218) based ceramic scaffolds, such as hydroxyapatite (HA) (Product No. 289396, 677418, 693863, etc.) and β-tricalcium phosphate (β-TCP) (Product No. 13204, 21218, and 49963)12 have been extensively studied and used in clinical applications.13 CaPs scaffolds, being a major constituent of bones, exhibit high osteoconductivity. They also present high compressive strength, which can even be improved using dopant additives such as SiO2 (Product No. 805890, 806587, 806765, etc.) or ZnO (Product No. 14439, 96479, etc.). But their processability is reduced and, therefore, possible geometries are limited. Metals are also used, usually titanium or stainless steel to ensure biocompatibility. They present a high mechanical strength but are non-biodegradable.13 Finally, composite materials have been developed by mixing two or more materials with the goal of combining the advantage of each individual material into one. One example is a polymer/ceramic composite, such as PCL/TCP or PCL/HA, in which the ceramic is integrated into the polymer to improve the mechanical integrity and bioactivity of the polymer.14 Composites show promising results for acellular scaffolds, with many potential combinations remaining to be explored.
Manufacturing Process
Numerous 3D printing and AM processes have been developed and commercialized since the 1980s,15,16 such as Stereolithography (SLA), Selective Laser Sintering (SLS), and Fused-deposition Modeling (FDM). These printing techniques can be used for the bioprinting of acellular scaffolds because they require less restrictive precautions like speed, temperature, toxicity and pressure for printing.
SLA consists of deflecting a laser beam in a horizontal plane to cure a photosensitive material in order to form a fixed layer.16 This layer is then moved along the vertical axis to allow the next adjoining layer to be created, as shown in Figure 2. This technology permits high resolution printing, with a layer thickness as small as 20 μm. In the horizontal plane, the resolution is defined by the diameter of the laser (around 250 μm); the use of Digital Light Projection (DLP) in place of a laser can improve the resolution to 70 μm. However, SLA limits the biochemical composition of the constructs to a single material, which must also be photosensitive.
SLS, in contrast, uses a high powered laser (Figure 2) to heat and fuse a powder-based material. By rastering the laser over the powder bed, the successive layers are fabricated. Once each layer is complete, another layer of powder is added to the top of the previous one, to be sintered by the laser to form the next layer. This is repeated until the entire part is produced.16 Scaffolds manufactured using SLS show high mechanical strength and shape complexity, since sintering provides better bonds between each layer and the presence of unsintered powder gives support for each successive layer. The resolution and the surface finish can vary depending on the powder.
3D printing using FDM consists of the positioning of an extruding nozzle in order to deposit strands of material in 3D space. The extrusion material is thermally melted inside the nozzle, solidifying after cooling upon deposition to create a layer (Figure 2). Materials used for FDM must exhibit a molten phase, making certain polymers and composites well-suited for this process. Since the process is strand-based, it is highly suitable for porous structures. However, complex geometries, such as overhanging layers, are difficult to manufacture.
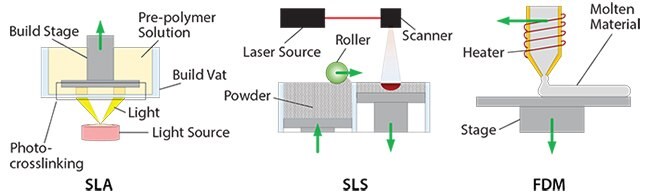
Figure 2.3D printing processes for the manufacturing of acellular scaffolds.
Manufacturing Soft Materials for Cell Encapsulation
Although acellular scaffolds can provide mechanical support and structural guidance for the growth of cells, post-processing cell seeding and/or biomolecule loading are required if cells and/or biomolecules need to be attached to the scaffold. This is a delicate task and does not allow for controlled attachment and spatial distribution of cells and biomolecules within the scaffold. However, loading is easier to achieve by encapsulating the cells and/or biomolecules directly in the printed material. By combining different cell types and growth factors according to designed biomimetic patterns, highly complex tissue constructs can be achieved.2 Such tissue constructs have many applications, allowing significant progress toward 3D miniature tissue models for drug delivery tests. To meet the needs of these demanding applications, however, sterile conditions, non-toxic materials, mild fabrication processes, and relatively short processing time windows are required, impacting both the choice of the material and the printing process.
Materials
Cell encapsulation requires the printing material have high water content and sufficient porosity to enable cells to receive nutrients and oxygen from the environment as well as remove waste in order to stay alive. The material should be soft and biodegradable to allow the cells to spread, migrate, proliferate, and interact with each other.17 The most commonly used materials for cell encapsulation are hydrogels, which can be either natural or synthetic. Natural hydrogels, such as gelatin and collagen, are extracted from animal or human tissues, presenting intrinsic molecular interactions with cells. Synthetic hydrogels, such as poly(ethylene glycol) or PEG, are widely used in bioprinting because of the flexibility of their physical properties. Depending on the gelation principle, hydrogels can be divided into two categories: physical and chemical hydrogels.15 A hydrogel is formed “physically” by changing the temperature, pH value, or other physical properties, while a “chemical” hydrogel is produced by crosslinking through covalent bonds. In the first case, the hydrogel can reverse back to its liquid state if the physical property is changed back to its initial state; whereas in the second case, the gelation is irreversible due to the water-insoluble network created by the covalent bonds formed. Chemical crosslinking is achieved by mixing two mutually reactive chemicals, while photocrosslinking is performed by exposing a solution composed of a photosensitive polymer and a photoinitiator to visible or UV light.16
Manufacturing Process
Inkjet-based bioprinting has been widely used to form 3D cell-laden constructs by continuously ejecting cell-laden droplets onto a destination stage using a thermal or an acoustic actuator. The nature of the print head designates the 3D construct be built dot by dot for each layer. Inkjetbased bioprinters are common for bioprinting applications because they have a fast printing speed, are compatibile with biological components, and are low cost. The viscosity of the printing material should be considered when choosing this printing method in order to reduce clogging the print head.
SLA can also be used to manufacture cell-laden constructs by adding cells to an uncrosslinked pre-polymer material. Visible light is the preferred light source for crosslinking when using DLP due to cell sensitivity to UV exposure and changes in temperature. While SLA offers high resolution, the tradeoff between printing quality and total processing time should be considered to yield optimal conditions for cell viability. SLA requires a larger amount of material than other bioprinting methods due to the requirement of filling the vat (Figure 3) with the printing material, which presents a major drawback if using expensive materials.
Another way to print hydrogel constructs is extrusion. To do this, a chamber is filled with cell-laden biomaterial, then using either pneumatic or piston-driven extrusion, the material is propelled through the print head. In order to create cell-laden constructs layer by layer, the print head robotically follows the desired path. For a physically formed hydrogel, the struts are extruded and gelled on stage upon change in pH, temperature, or other physical condition. Photocrosslinkable materials can also be used for this process. Once a layer of pre-polymer solution is extruded, it is crosslinked by exposure to light. Although the printing speed and the amount of extrusion can be precisely controlled, significant shear stress on the material can impact cell viability and should be carefully avoided.
Laser-based bioprinting is a commonly used technique in which a laser is used to transfer cell-laden materials from a source plate to the deposition stage. To achieve this transfer, the source plate is coated in a double layer with a laser-absorbing layer and a donor layer of biomaterial (Figure 3). When the laser pulse focuses on the laser-absorbing layer, the heated region generates a bubble to propel and deposit a droplet of biomaterial onto the destination stage. Use of a laser instead of a nozzle allows for the deposition of highly viscous materials with high accuracy. However, the heat generated by the laser hinders cell viability, and this process is the most limited in terms of vertical constructs.
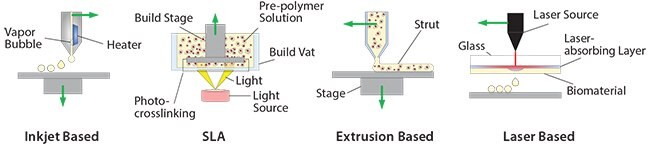
Figure 3.3D printing process for the manufacturing of soft materials for cell encapsulation.
Current Challenges and Perspectives
We have provided a brief overview of the current state of the art in bioprinting based on the two predominant approaches. In the first approach, acellular scaffolds are used to provide high resolution and highly reproducible implantable templates to promote cell function and tissue regeneration. In the second approach, cells are encapsulated directly in the material for integration inside a construct during printing. Materials used for this method contain a high ratio of water and are, thus, considered to be soft materials in terms of stiffness. While the first approach has been deliberately dedicated to stiffer materials in this section, it should be noted that acellular scaffolds based on soft materials can also be achieved using processes described in the earlier section “Manufacturing Soft Materials for Cell Encapsulation.”
While already showing very promising results, bioprinting is still in its early stage, and several challenges remain to be addressed to move the field forward. For example, as previously discussed, current 3D bioprinting approaches have limited capability for integrating the soft and rigid multifunctional components required for tissues and organs because of their inherent heterogeneity in mechanical, physical, chemical, and biological properties and functions. As a step toward this goal, our lab has developed a 3D hybrid bioprinter (Hybprinter) that can continuously and rapidly integrate cell-laden soft materials and rigid frame materials using FDM, SLA, and extrusion-based techniques in a controllable and automated manner under a single platform.18 By taking advantage of each process, we are able to manufacture constructs with both acellular scaffolds and cell-laden hydrogels, a step toward highly complex multimaterial constructs.
While bioprinting shows promise in tissue engineering, improvements in the printing processes are required. Increased speed should be considered in order to ease scale-up for acellular scaffolds and improve cell viability for cell-laden constructs. Moreover, higher resolution is required, in particular for the manufacturing of heterogeneous composite tissues and vascularized tissues. Vascularization is one of the key components and arguably the greatest challenge of tissue engineering.19 Such tissues consist of a highly complex vascular network, from millimeter-sized vessels to micrometer-scale capillaries. Reproducing such a network is a huge challenge, and up to now it has mainly been addressed by providing sufficient space in the porous scaffold for vascular tissues to spontaneously develop. A 3D printing process such as two-photon polymerization20 allows the manufacturing of parts with a micron-to-millimeter range of size and is, therefore, considered as a promising bioprinting process for the manufacturing of vascularized tissue constructs.
Research should also focus on the development of new materials that have improved biological properties and are suitable for bioprinting. Additionally, research should focus on using the current or future technologies to improve assembly of existing biomaterials to better mimic the complexity of the ECM, or the combination of both. Available materials for cell-laden tissue constructs dedicated to bioprinting are currently limited, but improved imaging capability and a fundamental currently limited, but improved imaging capability and a fundamental understanding of the complexity of tissues and developmental biology will contribute to the development of new materials and bioprinting technologies. It remains to be seen to what extent biomimetic complexity of the bioprinted constructs either in chemistry or in physical structure is necessary to achieve better healing and restoration of lost functions.
A growing number of AM applications are emerging, with bioprinting emerging as one of the most promising and challenging manufacturing processes due to the potential impact on global healthcare concerns like aging, organ transplantation, cancer therapy, and personalized and precision medicine. In the future, bioprinting has the potential to become both a source of miniature disease and toxicology models for the pharmaceutical industry and as a source of life-sized tissue/organ replacements for clinical treatments.
Acknowledgments
We would like to acknowledge the financial support of the following agencies: NIH R01AR057837 (NIAMS), NIH R01DE021468 (NIDCR), DOD W911NF-14-1-0545 (DURIP), DOD W81XWH-10-1-0966 (PRORP), and Stanford Coulter Translational Seed Grant.
Materials
References
To continue reading please sign in or create an account.
Don't Have An Account?