L-Asparagine in Cell Culture
What is Asparagine?
L-asparagine is an amino acid is an important component of mammalian proteins and is closely related to aspartic acid. Its name is derived from the vegetable asparagus, as the compound was first isolated from asparagus juice by French chemists Pierre Jean Robiquet and Louis Nicolas Vauquelin in 1806. L-asparagine is widely found in other plants.
L-asparagine is required by all cells for their protein synthesis and healthy growth but is classified as a nonessential amino acid. This is because normal mammalian cells obtain most of the asparagine they need through their own synthesis.
Related Products
Response not successful: Received status code 500
Cancer cells need an increased quantity of L-asparagine when compared to non-cancerous cells. This is to cope with their faster growth and proliferation than normal cells. However, most cancer cells cannot produce enough of the amino acid by themselves.
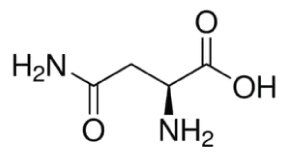
Figure 1.Structure of L-asparagine
Function of L-asparagine in cells
Mammals can synthesize L-asparagine from aspartic acid with the action of asparagine synthetase (EC 6.3.5.4). The amide group of the amino acid is derived through a reaction between aspartate and glutamine. This reaction occurs in the presence of energy in the form of ATP and yields asparagine and glutamate (Figure 2). This reaction is reversible, so asparagine can be converted to aspartic acid.
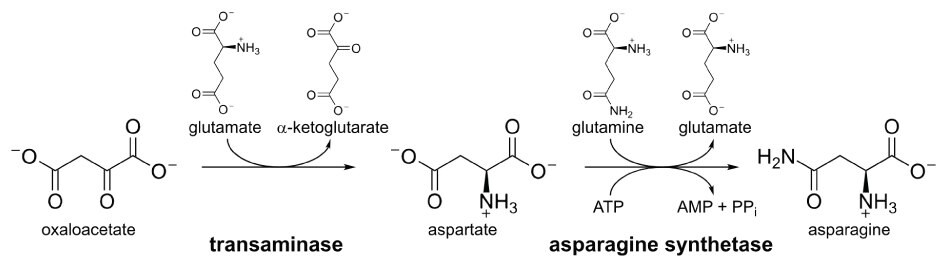
Figure 2.Biosynthesis of asparagine.
The conversion of asparagine into aspartate by asparaginase (EC 3.1.1.5) is important in the cell, as aspartate plays critical roles in multiple cellular functions. This includes the citric acid cycle, urea cycle and neural signal transmission. L-asparagine also acts as a precursor for several amino acids, including arginine.
Although L-asparagine can be obtained from food intake, the molecule cannot freely pass through the blood-brain barrier to enter the brain. This is important, as the amino acid is precursor of aspartic acid, a neurotransmitter and neuromodulator1 . Therefore, brain cells mainly depend on local asparagine synthesis2.
Because it is an amino acid, L-asparagine is a component of many proteins. In cellular proteins, the terminal amine group of the carboxamide side chain of L-asparagine makes efficient hydrogen bond interactions with the peptide backbone. Therefore, it is often found near the ends of alpha-helices and in turn motifs in beta sheets.
The carboxamide side chain of L-asparagine is also a key site for N-linked glycosylation. This post-translational modification is found in proteins and can influence their properties and bioactivities.
L-asparagine in cell culture
L-asparagine is a supplement that is often used in cell culture media to promote cell growth. It is one of the components in MEM non-essential amino acids solution. L-asparagine is also an important supplement in Dulbecco′s modified Eagle′s medium (DMEM) when culturing various primary cell lines.
L-asparagine can enhance the activity of ornithine decarboxylase (EC 4.1.1.17) in cultured IEC-6 intestinal epithelial cells and in cultured human colon adenocarcinoma Caco-2 cells when added as a supplement to the cell culture media. It has also been used as a component of LHC basal media for the culture of human airway epithelial cells.
Certain types of leukemic cells and NK/T cell lymphoma lack of the ability to synthesize asparagine due to a deficiency in L-asparagine synthetase (ASNS, EC 6.3.5.4). Therefore, these cells need external L-asparagine supplementation. L-asparagine has been found playing critical roles in tumor cell growth3 . It was shown to be an important supplement for solid tumors, including breast cancer, non-small-cell lung cancer (NSCLC), prostate cancer, and pancreatic tumors.
Multiple engineered Chinese hamster ovary (CHO) cell strains have been widely used to produce various recombinant proteins, such as monoclonal antibodies (mAb), for therapeutic applications. Data suggest that optimized usage of L-asparagine in the culture media will enhance the yield of bioproducts in CHO cell culture dramatically.
By supplementing CHO culture media with a ratio of asparagine and the amino acid glutamine, the antibody production and specific antibody production rate can be improved4,5. When supplementing culture media with asparagine and with another amino acid, cysteine, cell-specific productivity in CHO cells increased by 85%6.
L-asparagine has applications other than human cell culture. For instance, in the presence of L-asparagine, spore germination in Bacillus subtilis increases. Asparagine is also the storage form for aspartic acid. L-aspartic acid is a non-essential amino acid that is formed from the deamination of asparagine, and is one of the components in synthetic dropout media when culturing budding yeast. In addition to budding yeast culture, it is commonly used to study non-enzymatic gluconeogenesis.
Chemical characteristics and stability of asparagine
Asparagine has the molecular formula of C4H8N2O3 and molecular weight of 132.12 g/mol. It is polar amino acid containing an uncharged side chain, with an isoelectric point (pH) of 5.41 and pka equal to 2.02 and 8.80.
It can be deaminated non-enzymatically into ammonia and aspartic acid in cell culture medium. Therefore, it should be added to the culture medium just before use to reduce the accumulation of toxic free ammonium ions7.
References
To continue reading please sign in or create an account.
Don't Have An Account?