Quantification of Bitter Acids in Hops by Thin Layer Chromatography
Introduction
The amount of bitter acids in hops is a very important parameter for beer production. Different types of hops have different quantities of alpha and beta acids, which are responsible for the bitterness of the beer. In principle the hops are divided into aromatic hops (<10% of alpha acids) and bitter hops (>10% alpha acids).
A total of 12 samples were analyzed. Four were from different aromatic hops, four were from bitter hops, and the last four samples were the same variety of hops from different regions. All of the samples were applied on HPTLC MS-grade plates and developed with the AMD 2 procedure. After development the plates must be dried for 5 minutes. Then they can be directly scanned without any additional preparation steps. The advantage of TLC is that lots of samples can be applied on one plate, developed in one step and analyzed together, saving time.
Results and Discussion
Using HPTLC MS-grade plates and the AMD 2, it was possible to separate the alpha (Rf 0.36) and beta acids (Rf 0.65) (marked with orange lines) in hops from the other substances (Figure 1). In the next step the acids were quantified, using the International Calibration Extract.
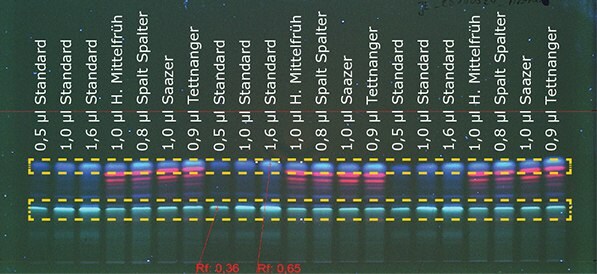
Figure 1A.Detection of aromatic hop at 366 nm
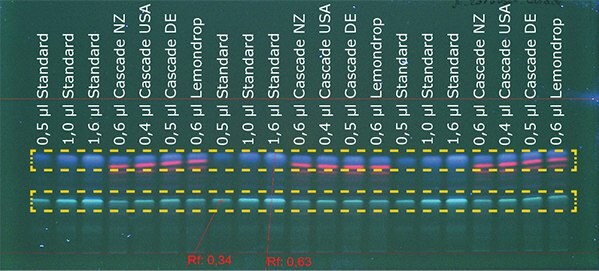
Figure 1B.Detection of cascade hop at 366 nm

Figure 1C.Detection of bitter hop at 366 nm
The standard was applied with volumes between 0.5 μL and 1.6 μL. The different hop samples were applied using volumes between 0.3 μL and 1.0 μL. Details about sample preparation and chromatographic data can be found on page 3. The acid contents of the samples were calculated by means of a calibration curve (polynomial) using winCATS (Camag). Alpha and beta acid contents of the samples are shown in Table 1.
It is known that bitter hops have much more alpha acids than aromatic hops. However, it was not known whether beta acid content correlates with that of alpha acids. By comparing four of the same variety of hop of different origins, this analysis can also help to determine if the bitter acid content is region dependent.
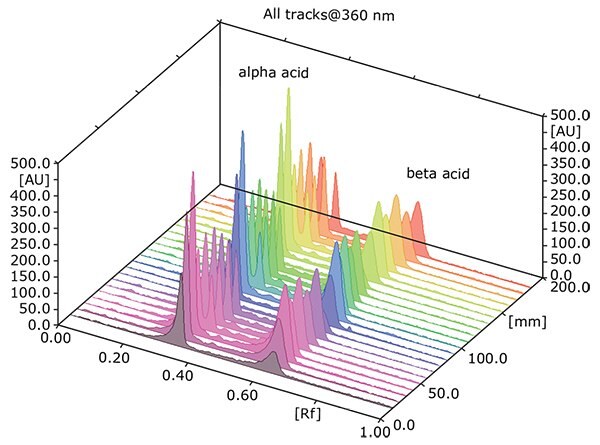
Figure 2: Scan of the aromatic hops as an example for all hop samples at 360 nm.
Conclusion
By the use of HPTLC the difference of the bitter acid content in various hop samples can be shown. Although hops contain a high amount of matrix, both types of bitter acids can be separated from other substances. Furthermore the acids can be quantified by scanning under UV light. This study allowed both regional and varietal differences of bitter acid content in hops to be determined. In conclusion, the bitter hop samples have much more bitter acids than the aromatic hop samples. The beta acid content is not dependent on variety or region and is quite similar in all hop samples.
Determined bitter acid values | ||
|---|---|---|
| Compound | α-acid [%] | β-acid [%] |
| Mittelfrüh Hallertau | 2.74 | 2.98 |
| Spalt Spalter | 3.83 | 5.44 |
| Saazer | 3.18 | 2.82 |
| Tettnanger | 3.12 | 3.61 |
| Cascade NZ | 4 | 3.86 |
| Cascade USA | 8.06 | 8.04 |
| Cascade Hallertau | 6.45 | 6.06 |
| Lemondrop USA | 4.11 | 4.2 |
| Apollo | 13.48 | 5.81 |
| Green Bullet | 9.05 | 5.95 |
| Hallertau Herkules | 3.98 | 3.32 |
| Topaz | 16.2 | 7.22 |
Chromatographic Conditions | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Chromatography | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Plate | HPTLC Silica gel 60 F254 MS-grade, 20x10 cm | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Standard | 1 mg/mL of the International Calibration Extract 3 (contains 44.64% alpha acid, 24.28% beta acid and 31.08% other ingredients) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sample Preparation | Crumbled the hop-pellets (5 g), added 10.0 mL MeOH, 50.0 mL diethyl ether and 20 mL 0.1 molar hydrochloric acid solution. The solution stirred for 40 min, 2 mL of the upper ether phase removed, the residue filled in a 20.0 mL volumetric flask and filled up with MeOH. The extract filtered with a 0.45 μm syringe filter. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sample Application | ATS 4 automatic TLC sampler (Camag), 6 mm bandwise | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Application Volume | 0.3 – 1.6 μL | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Mobile Phase | Ethyl acetate/Methanol/n-Heptane as a gradient, developed with the AMD 2 automated multiple development (Camag)
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Migration Distance | 5 cm | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Migration Time | 55 min (complete AMD method) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Documentation Equipment | Reprostar 3 (Camag) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Detection | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Wavelength | UV light at 366 nm | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Scan Equipment | TLC Scanner 3 (Camag) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Wavelength | UV light at 360 nm | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Chromatographic data | |||||
|---|---|---|---|---|---|
| Category | Compound | Concentration | Application volume | Rf value α-acid | Rf value β-acid |
| Aromatic hops | ICE standard | 1 mg/mL | 0.5 μL | 0.35 | 0.65 |
| ICE standard | 1 mg/mL | 1.0 μL | 0.35 | 0.65 | |
| ICE standard | 1 mg/mL | 1.6 μL | 0.35 | 0.65 | |
| Mittelfrüh Hallertau | – | 1.0 μL | 0.35 | 0.65 | |
| Spalt Spalter | – | 0.8 μL | 0.35 | 0.65 | |
| Saazer | – | 1.0 μL | 0.35 | 0.65 | |
| Tettnanger | – | 0.9 μL | 0.35 | 0.65 | |
| Same hop of different regions | Cascade NZ | – | 0.6 μL | 0.35 | 0.65 |
| Cascade USA | – | 0.4 μL | 0.35 | 0.65 | |
| Cascade Hallertau | – | 0.5 μL | 0.35 | 0.65 | |
| Cascade Lemondrop USA | – | 0.6 μL | 0.35 | 0.65 | |
| Bitter hops | Apollo | – | 0.3 μL | 0.35 | 0.65 |
| Green Bullet | – | 0.5 μL | 0.35 | 0.65 | |
| Hallertau Herkules | – | 0.8 μL | 0.35 | 0.65 | |
| Topaz | – | 0.3 μL | 0.35 | 0.65 | |
Materials
Response not successful: Received status code 500
To continue reading please sign in or create an account.
Don't Have An Account?