Sign In to View Organizational & Contract Pricing.
Select a Size
Change View
| Pack Size | SKU | Availability | Price |
|---|---|---|---|
| 50 mg | Please contact Customer Service for Availability | $997.00 |
About This Item
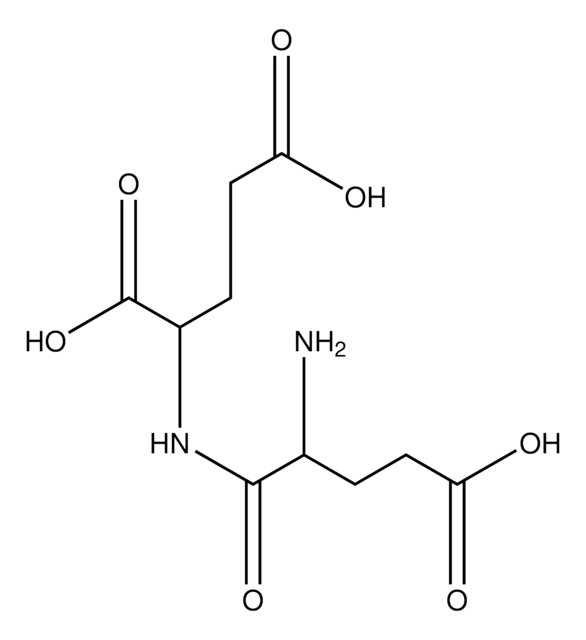
Empirical Formula (Hill Notation):
C5H9NO5
CAS Number:
Molecular Weight:
163.13
NACRES:
NA.28
PubChem Substance ID:
UNSPSC Code:
12352202
MDL number:
Beilstein/REAXYS Number:
1725871
Product Name
(4R)-4-Hydroxy-L-glutamic acid, ≥98.0% (TLC)
Quality Level
assay
≥98.0% (TLC)
form
powder
optical activity
[α]/D 20.5±1.5°, c = 1 in H2O
color
white
mp
171 °C
storage temp.
−20°C
SMILES string
N[C@@H](C[C@@H](O)C(O)=O)C(O)=O
InChI
1S/C5H9NO5/c6-2(4(8)9)1-3(7)5(10)11/h2-3,7H,1,6H2,(H,8,9)(H,10,11)/t2-,3+/m0/s1
InChI key
HBDWQSHEVMSFGY-STHAYSLISA-N
Biochem/physiol Actions
(4R)-4-Hydroxy-L-glutamic acid or (2S,4R)-4-hydroxyglutamate was shown to activate the metabotropic glutamate receptors, mGlu1a, mGlu2, and mGlu8a in a dose-dependent manner. [1]
Packaging
Bottomless glass bottle. Contents are inside inserted fused cone.
1 of 1
This Item | |||
|---|---|---|---|
| form powder | form powder | form powder | form powder |
| assay ≥98.0% (TLC) | assay ≥98.0% (TLC) | assay ≥98% (TLC) | assay ≥98% (HPLC) |
| storage temp. −20°C | storage temp. 2-8°C | storage temp. −20°C | storage temp. 2-8°C |
| Quality Level 100 | Quality Level 200 | Quality Level 200 | Quality Level 200 |
| color white | color white | color white | color - |
| optical activity [α]/D 20.5±1.5°, c = 1 in H2O | optical activity - | optical activity - | optical activity - |
Storage Class
11 - Combustible Solids
flash_point_f
Not applicable
flash_point_c
Not applicable
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Metabolism of gamma-hydroxyglutamic acid. I. Conversion to alpha-hydroxy-gamma-ketoglutarate by purified glutamic-aspartic transaminase to rat liver.
A GOLDSTONE et al.
The Journal of biological chemistry, 237, 3476-3485 (1962-11-01)
Lennart Bunch et al.
ChemMedChem, 4(11), 1925-1929 (2009-09-05)
Subtype-selective ligands are of great interest to the scientific community, as they provide a tool for investigating the function of one receptor or transporter subtype when functioning in its native environment. Several 4-substituted (S)-glutamate (Glu) analogues were synthesized, and altogether
A S Bessis et al.
Bioorganic & medicinal chemistry letters, 11(12), 1569-1572 (2001-06-20)
The (2S,4R)- and (2S,4S)-4-hydroxyglutamates activate cloned mGlu(1a), mGlu(2), and mGlu(8a) receptors with different potencies. Best results were obtained with the (2S,4S) isomer being almost as potent as glutamate on mGlu(1a)R and mGlu(8a)R. Data are interpreted on the basis of the
Global Trade Item Number
| SKU | GTIN |
|---|---|
| 76157-50MG | 04061832720265 |
| 76157-10MG | 04061832720258 |