Select a Size
| Pack Size | SKU | Availability | Price |
|---|---|---|---|
| 5 mg | Please contact Customer Service for Availability | $229.00 | |
| 10 mg | Please contact Customer Service for Availability | $398.00 | |
| 25 mg | Please contact Customer Service for Availability | $784.00 |
About This Item
biological source
synthetic (organic)
Quality Level
assay
≥93% (TLC)
form
powder
optical activity
[α]20/D 40.8 to 51.0 °, c = 0.66% (w/v) in water
color
white
solubility
H2O: soluble 50 mg/mL, clear, colorless
storage temp.
−20°C
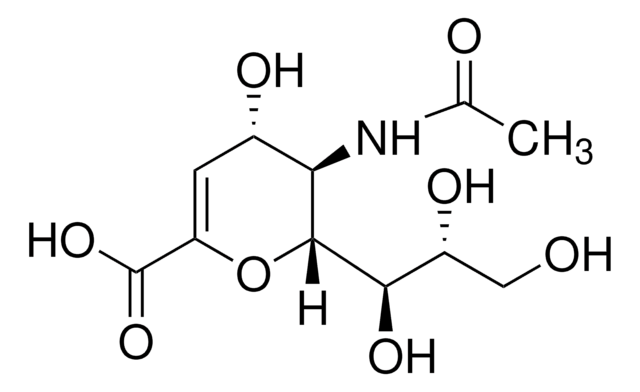
SMILES string
CC(=O)N[C@@H]1[C@@H](O)C=C(O[C@H]1[C@H](O)[C@H](O)CO)C(O)=O
InChI
1S/C11H17NO8/c1-4(14)12-8-5(15)2-7(11(18)19)20-10(8)9(17)6(16)3-13/h2,5-6,8-10,13,15-17H,3H2,1H3,(H,12,14)(H,18,19)/t5-,6+,8+,9+,10+/m0/s1
InChI key
JINJZWSZQKHCIP-UFGQHTETSA-N
Application
Biochem/physiol Actions
Other Notes
1 of 1
This Item | |||
|---|---|---|---|
| assay ≥93% (TLC) | assay ≥98% (HPLC) | assay ≥98% (titration) | assay ≥93% (HPLC) |
| biological source synthetic (organic) | biological source - | biological source synthetic (organic) | biological source - |
| solubility H2O: soluble 50 mg/mL, clear, colorless | solubility water: ≥10 mg/mL | solubility 1 M HCl: 50 mg/mL, clear, colorless | solubility water: 50 mg/mL, clear to slightly hazy, colorless to faintly yellow |
| Quality Level 300 | Quality Level 100 | Quality Level 300 | Quality Level 200 |
| form powder | form solid | form powder | form powder |
| storage temp. −20°C | storage temp. −20°C | storage temp. room temp | storage temp. −20°C |
Storage Class
11 - Combustible Solids
wgk
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type N95 (US)
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Global Trade Item Number
| SKU | GTIN |
|---|---|
| D9050-5MG | 04061833590188 |
| D9050-10MG | 04061833590171 |
| D9050-25MG | 04061832389240 |